My first draft of this got lost in the computer (Thanks, all you coders who think you know how to create intuitive, ease to use interfaces). So here’s another attempt to get stuff from one location to another. We’ll see how it it goes…
A while back I posted a Powerpoint presentation on research on music, dance, and singing, which cited a review in the Movement Disorders Society (2018, I believe), in which the conclusion was that exercise did not affect Parkinson’s Disease (PD) symptoms. Since then, there have been many more studies on the effects of different modalities of exercise. In this first article, a meta review of publications, found that exercise is good for you if have PD.
The purpose of the review was to compare and rank the types of exercise that improve PD symptoms by quantifying information from randomised controlled trials. Out of 10,474 citations, 250 studies were included involving 13,011 participants. The results (drumroll, please):
- Power training (PT) had the best benefits for motor symptoms
- Body weight support treadmill training showed the best improvement in balance, gait velocity and walking distance
- Robotic assisted gait training had the most benefits for freezing of gait
- Dance showed the best benefits for depression
- Only Yoga significantly reduced anxiety symptom compared with controls
- Only resistance training significantly enhanced sleep quality and cognition
- Physical Therapy showed the best results for muscle strength
- Five types of therapy showed improvement in concerns about falling versus control groups.
If interested, the full free text is available at the linked citation above. Other reviews have shown different results. Best recommendation is to find a variety of exercises that you enjoy, and do them as often as you can. But don’t try so hard that you wind up hurting yourself.
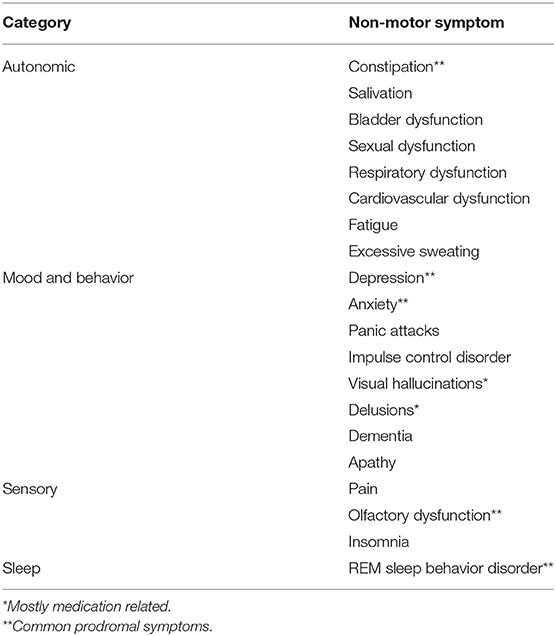
The second meta review alluded to in the title has to do with sleep disorders.
Fifty-three longitudinal studies of sleep in PD were identified, which included:
- Excessive daytime sleepiness,
- Insomnia,
- Obstructive sleep apnea,
- Rapid eye movement sleep behavior disorder (RBD),
- Restless legs syndrome, and
- Shift work disorder were studied in addition to other studies that had focused on either multiple sleep disorders or broadly on sleep disorders as a whole.
RBD is now considered an established prodromal feature of PD, but other sleep disorders do not clearly increase risk of subsequent PD.
I’ll throw in one last citation. Not just because it plays on the title to one of Bob Dylan’s songs, but also because it is one of many nutritional supplements prescribed to aid in cleaning out neural cell debris in (where else?) Bob’s Brain:
and that ends this day.